Frequently asked questions (FAQs) about mesenchymal stem cell therapy:
Table of Contents
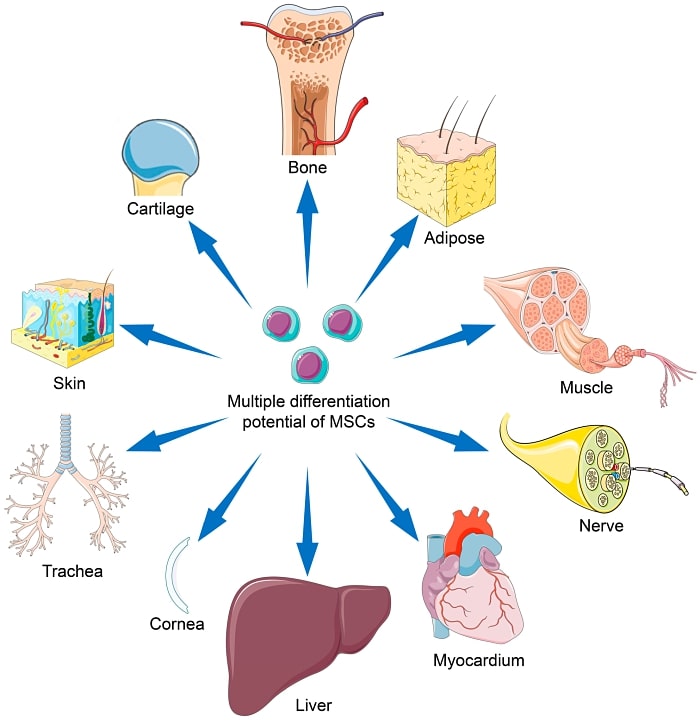
What are mesenchymal stem cells (MSCs)?
Mesenchymal stem cells (MSCs) are multipotent cells that can differentiate into various cell types, including bone cells, cartilage cells, and fat cells. They are found in various tissues such as bone marrow, adipose tissue, and umbilical cord.
How does mesenchymal stem cell therapy work?
MSC therapy involves the isolation of mesenchymal stem cells from a donor source, followed by their injection into a patient. The cells have the potential to differentiate into specific cell types and may also release signaling molecules that promote tissue repair and modulate the immune response.
What conditions can be treated with mesenchymal stem cell therapy?
MSC therapy is being researched and used for various conditions, including orthopedic injuries, autoimmune diseases, neurodegenerative disorders, and cardiovascular diseases. However, its efficacy is still being studied, and not all applications have been proven.
Is mesenchymal stem cell therapy FDA-approved?
As of my last knowledge update in January 2022, the FDA has approved certain mesenchymal stem cell therapies for specific medical conditions. However, the regulatory landscape may change, so it’s essential to check for the latest information.
Are there any risks or side effects associated with mesenchymal stem cell therapy?
While MSC therapy is generally considered safe, potential risks include infection, an immune response to the cells, or unintended differentiation into undesired cell types. The long-term effects and safety are still areas of active research.
Where do mesenchymal stem cells come from for therapy?
Mesenchymal stem cells can be derived from various sources, such as bone marrow, adipose tissue (fat), umbilical cord tissue, and placenta. The choice of the source depends on factors like accessibility, cell yield, and ethical considerations.
How are mesenchymal stem cells harvested and prepared for therapy?
The process involves isolating the mesenchymal stem cells from the chosen tissue source, followed by their expansion in culture. The cells are then prepared for injection, which may involve purification and quality control measures.
What is the difference between autologous and allogeneic mesenchymal stem cell therapy?
Autologous therapy uses a patient’s own stem cells, typically harvested from their bone marrow or adipose tissue. Allogeneic therapy involves using stem cells from a donor, which are carefully matched to reduce the risk of rejection.
Is mesenchymal stem cell therapy covered by insurance?
In many cases, mesenchymal stem cell therapy is not currently covered by insurance because it is considered experimental or investigational for many conditions. Patients should check with their insurance providers for the most accurate and up-to-date information.
What is the current state of research on mesenchymal stem cell therapy?
Research on MSC therapy is ongoing, with numerous clinical trials exploring its potential for different medical conditions. While some promising results have been reported, more research is needed to establish its safety, efficacy, and long-term effects for various applications.
Why Choose Us ?
As one of the Leading Aesthetic Clinic in Malaysia, NextMed Clinic provides top-notch procedures by Certified Medical Practitioners. Whatever your concerns, be it enhancing your natural features or reversing the signs of aging, NextMed Clinic is able to deliver a wide range of quality, state of art & personalized aesthetic, general health services & wellness solution, merged with exemplary service, in a warm and pleasent enviroment designed to make you feel completely at ease.
At NextMed Clinic, we strongly believe that each patient is unique. Therefore treatments are customized to each patient’s specific needs.

Certified Doctor
.